Function Annotation for BL18491
| Ligand ID | Name | Synonyms | Formula | 2D structure |
|---|---|---|---|---|
| U5P | Ump | "C9 H13 N2 O9 P" | 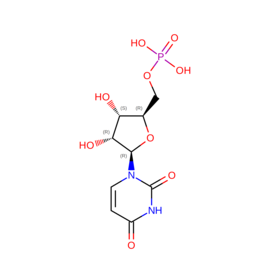 |
Binding affinity
| Experimental affinity | N/A | |||
| Predicted affinity | N/A, |
|||
Surface area (A2)
| Surface area of receptor | N/A |
| Surface area of ligand | N/A |
| Surface area of complex | N/A |
| Interface area | N/A |
Reference